Waterlase Trial Program
Experience the future of dentistry.
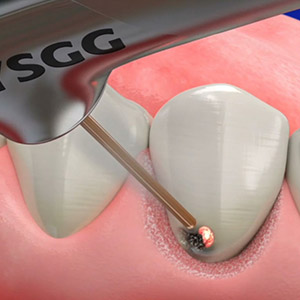
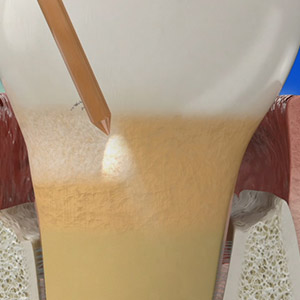
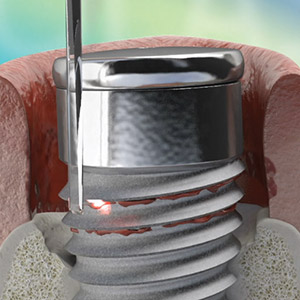
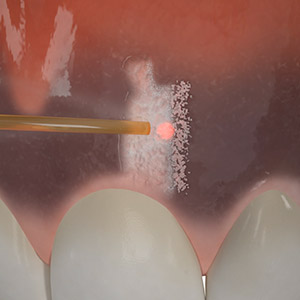
Test drive the Waterlase all-tissue laser in your office – at no charge. Start with a two-day hands-on training led by an experienced Waterlase clinician who will guide your education journey and provide support for a customized implementation program. Discover the potential of Waterlase and elevate your dental practice to new heights with the Waterlase Trial Program.
Learn more about this opportunity and how you can get started!